Request a representative visit or G7 15 Day or G7 samples
DIaMonD - A Randomized Controlled Trial Examining the Benefit of CGM Use for Adults with Type 1 Diabetes on Insulin Injections
DIaMonD Study1 shows that Dexcom CGM use improves glucose control in adults with type 1 diabetes using insulin injections.
DIAMOND TYPE 1 STUDY HIGHLIGHTS (PDF)
Background
Clinical trials demonstrating improved glucose control in patients with type 1 diabetes (T1D) using CGM have historically and predominantly involved participants who use insulin pump therapy and not insulin injections (MDI).2 However, most adult patients with T1D deliver insulin by injection.3
Objective
Examine effectiveness of CGM vs. self-monitoring of blood glucose (SMBG) with blood glucose meter in adults with T1D on MDI insulin therapy.
Primary endpoint: Change in A1C
Research Design/Methods
24-week, randomized, controlled trial of 158 adults with T1D. Study conducted using the Dexcom G4 PLATINUM CGM System, which uses the same software 505 as the Dexcom G5 Mobile CGM System.
- Group 1: MDI + CGM (n=105)
- Group 2: MDI + SMBG (n=53)
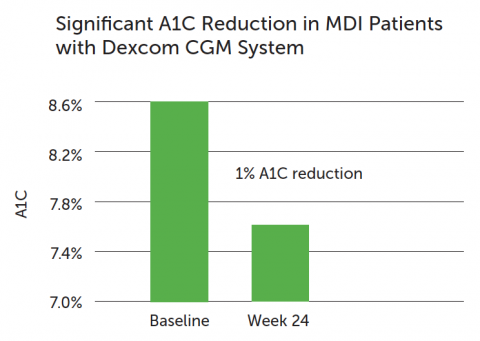
- Baseline A1C was 8.6% (range 7.5 to 9.9%)
- Various education (with 45% less than a bachelor’s degree) and numeracy levels (ability to use math in daily diabetes management decisions)
Patient Education/Touchpoints
All participants received the same basic diabetes education. Intensive diabetes education and device training was not a part of this study. Study was six months in length and used a protocol with minimal “touch,” reflecting clinical practice.
Multicentered
The trial was conducted at 24 endocrinology practices in the USA (19 community-based and 5 academic centers).
Results
Dexcom CGM use resulted in a significant A1C reduction in patients with T1D on insulin injections.
In study participants using Dexcom CGM:
Overall: an average 1% A1C reduction after 24 weeks of regular use, compared to baseline (difference of 0.6% compared to SMBG group [p-value <.001]). This included:
- ≥1% A1C reduction for 52% of patients
- Participants with a baseline A1C of ≥8.5% achieved an average of 1.3% A1C reduction compared to baseline
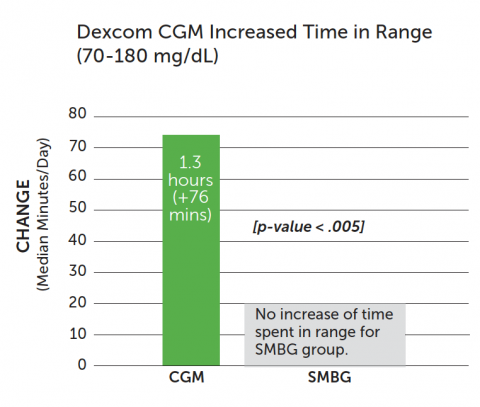
Dexcom CGM increased time spent in range from baseline (70 - 180 mg/dL):
- CGM group: median 1.3 additional hours (+76 mins) in range
- SMBG group: no increase of time in range
- CGM group achieved a 4% reduction in glycemic variability compared to SMBG group
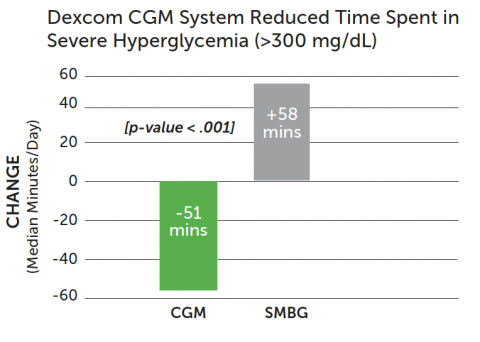
Dexcom CGM use reduced time in hyperglycemia.
- CGM group: 40% reduction of time spent in severe hyperglycemia (>300 mg/dL)*
- SMBG group: median increase of 58 minutes spent in severe hyperglycemia
Dexcom CGM use reduced time in hypoglycemia:
- 37 less minutes/day <70 mg/dL; 49% reduction from baseline
- 20 less minutes/day <60 mg/dL; 53% reduction from baseline
- 14 less minutes/day <50 mg/dL; 69% reduction from baseline
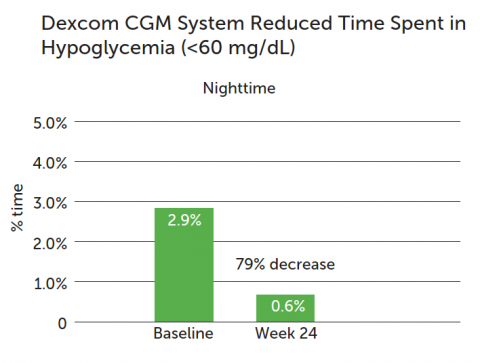
Dexcom CGM use reduced nighttime hypoglycemia (<60 mg/dL):
- CGM group: 79% reduction in the median time spent in hypoglycemia at night, from 2.9 to 0.6%.
CGM Use and Adherence
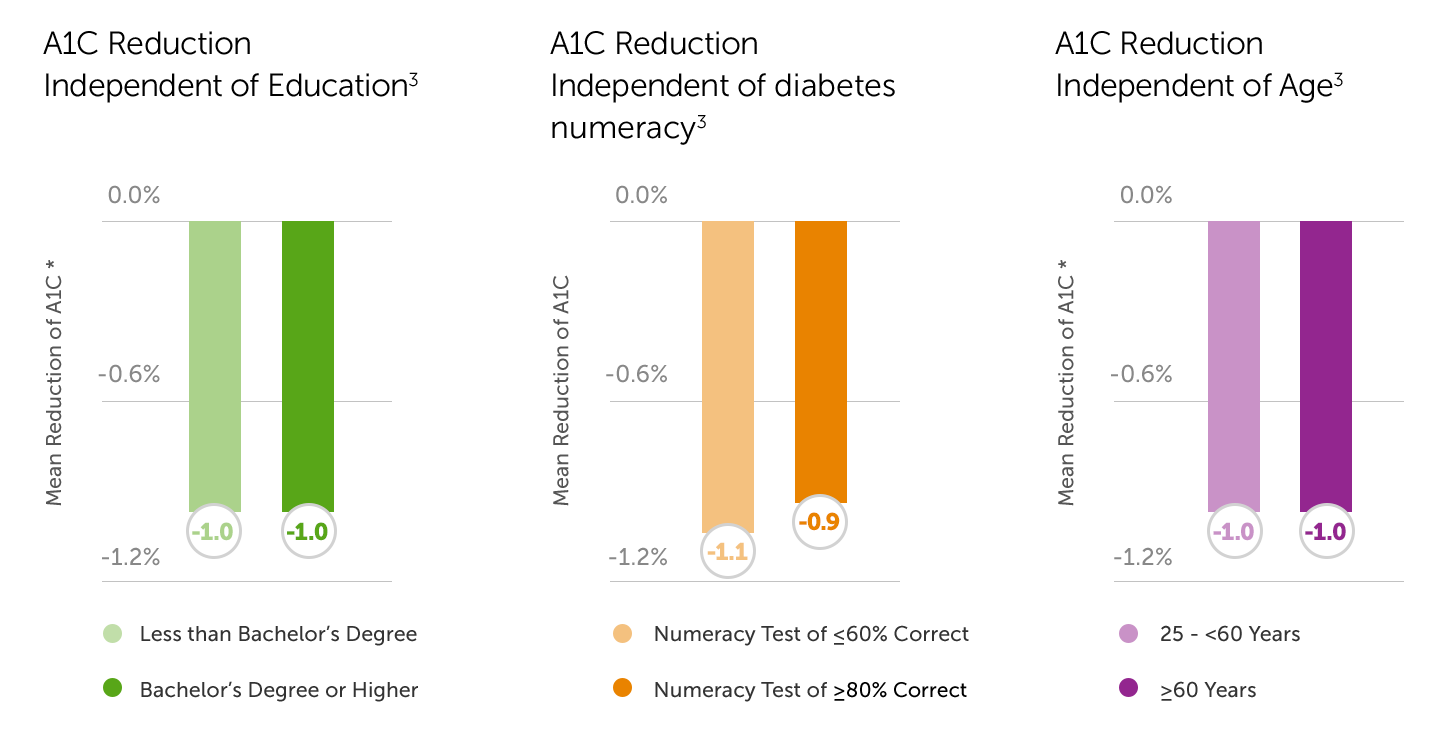
- A1C reductions observed with CGM use occurred independent of age, education, and diabetes numeracy.
- 93% of patients were using the Dexcom CGM ≥ 6 days/week (at week 24).
*Investigators pooled data from weeks 12 and 24; Δ time (median minutes/day): [pooled data - baseline data]
DIAMOND TYPE 1 STUDY HIGHLIGHTS (PDF)
1 Beck RW, Riddlesworth T, Ruedy K, et al. Effect of Continuous Glucose Monitoring on Glycemic Control in Adults With Type 1 Diabetes Using Insulin Injections: The DIAMOND Randomized Clinical Trial. JAMA. 2017;317(4):371–378. doi:10.1001/jama.2016.19975. View full article. [text link: https://jamanetwork.com/journals/jama/fullarticle/2598770]
2 Bergenstal RM, Tamborlane WV, Ahmann A, et al; STAR 3 Study Group. Effectiveness of sensor-augmented insulin-pump therapy in type 1 diabetes. N Engl J Med. 2010;363(4):311-320.
3 Grunberger G, Abelseth JM, Bailey TS, et al. Consensus statement by the American Association of Clinical Endocrinologists/American College of Endocrinology Insulin Pump Management Task Force. Endocr Pract. 2014;20(5):463-489.
Related Content
Topics: Pregnancy, Accuracy, Safety Research Spotlight: “Accuracy and Safety of Dexcom G7 CGM Use in Pregnancy Patients with Diabetes” Polsky S, et al. Diabetes Technol Ther. 2024;26(5):307-12...
Title: Long-Term Improvements in Glycemic Control with Dexcom CGM Use in Adults with Noninsulin-Treated Type 2 Diabetes Topics: Long-Term Outcomes, Type 2 Diabetes, Noninsulin-Treated Research...

