Request a representative visit or G7 15 Day or G7 samples
CSS [DO NOT REMOVE] [Hidden on page]
Our smartest technology delivers superior accuracy on day 1, for early, actionable data†,‡,2
Builds upon the G7 features you expect, now with additional enhancements.

Longest wear time of any CGM
15.5 days of wear (including a 12-hour grace period)†,§,3,4

Fast onboarding, more intuitive login
Getting started is easier than ever, and patients can now use facial recognition or thumbprint to log in

The most accurate glucose readings
Unrivaled accuracy so you can prescribe with confidenceII,2,5
Convenient meal and event tracking
Snap photos that track activities, meals, and medications, including GLP-1 agonists

98% Say it's easy to use
98% of users said Dexcom G7 15 Day is easy to use2
G7 15 Day CGM: a highly flexible and customizable experience

84% of CGM users
switching to Dexcom CGM report improved confidence in their diabetes management¶,6

Freedom to choose
Connects with major automated insulin delivery (AID) pumps, pens, and many popular lifestyle apps

Information where you want it
Review simplified patient data in-app or via Clarity during clinic visits
With G7 15 Day CGM, everyone wins
Harness the data that drives proven results

Accuracy that leads to outcomes
Industry-leading MARD of 8.0% increases confidence to help improve outcomes1,2,7-12

Smarter sensor technology
Improved accuracy designed to deliver better outcomes*,1,2,7-12

Useful insights into their activity
Easier patient journaling optimizes their entries and your insights

G7 series features, enhanced
Features you know and love from G7 including “Urgent Low Soon” and other alerts to match their needs
Better outcomes are a win for everyone


Noteworthy news
Stay informed with the latest updates on interoperability and coverage for Dexcom products.
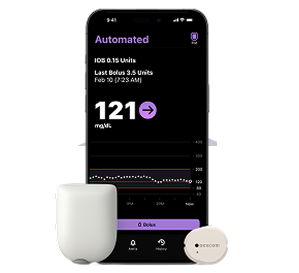
Control Omnipod® 5 and Dexcom G7 with your iPhone. Easily manage diabetes with the Dexcom G7 CGM system and Omnipod 5 automated insulin delivery system. The latest iOS update lets you control both from your iPhone.** Download the Omnipod 5 app to get started.
**Smartphone sold separately.
Pod shown without the necessary adhesive.

Dexcom G7 is the first and only CGM with direct connectivity to Apple Watch, giving patients actionable alerts and accurate glucose numbers8,9 with just a glance at their wrist. Now your patients can confidently leave their iPhone behind.||
Now you can share the same clinic code with all patients on Dexcom CGM:
- eliminates the need to send a unique code to each patient
- allows you to create a custom code for your clinic
- streamlines your clinic’s workflow
CGM=continuous glucose monitoring; CPT=Current Procedural Terminology; DME=durable medical equipment; GLP-1=glucagon-like peptide-1; MARD=mean absolute relative difference.
*Per clinical studies, not all sensors may last 15 days. †A study was conducted to assess the sensor life where 73.9% of sensors lasted the full 15 days. In other words, when using the product per the package labeling, approximately 26% of sensors may not last for the full 15 days. ‡Fingersticks required for diabetes treatment decisions if symptoms or expectations do not match readings.§Excludes implantable CGM systems. IIDexcom G7 15 Day is NOT indicated for pediatric patients.¶Based on data obtained from a survey of patients using a Dexcom CGM system not available in the United States. #Individual prices may vary depending on insurance coverage. Under Medicare’s DME fee schedule, reimbursement and coinsurance for CGM’s using CPT codes A4239 and E2103, are the same, regardless of CGM brand. **Smartphone sold separately.
1 Dexcom, (VV-07576) Data on File, 2025. 2 Garg SK, et al. Diabetes Technol Ther. 2025;27(6):413-502. 3 Dexcom G7 15 Day User Guide. 4 Data on file (VV-07558), Dexcom; 2025. 5 Data on file (VV-07560). Dexcom; 2025. 6 Data on file (VV-07597). Dexcom; 2025. 7 Beck RW, et al. JAMA. 2017;317(4):371-378. 8 Beck RW, et al. Ann Intern Med. 2017;167(6):365-374. 9 Martens T, et al. JAMA. 2021;325(22):2262-2272. 10 Welsh JB, et al. J Diabetes Sci Technol. 2024;18(1):143-147. 11 Clark TL, et al. Diabetes Technol Ther. 2024;26(10):700-708. 12 Ehrhardt N, et al. Clin Diabetes. 2020:38(2):126-131.
MAT-11234



